-
About Us
Keith J. Stine, Chair
Mission Statement
Degree Offerings
Department Photos
Outreach Activities
-
Undergraduate Studies
Undergraduate Degrees
Undergraduate Program Advising
Undergraduate Scholarships and Awards
Research for Undergraduates
Resources
-
Graduate Studies
Graduate Program Overview
Graduate Degrees
Application to Graduate Program
Graduate Awards
Graduate Program Contact Information
-
Faculty
Faculty Contact Information
Inorganic
Organic
Analytical/Physical
Biochemistry
Chemistry Faculty Awards
-
Staff
-
Seminar Programs
Archive
Graduate Student Seminars
Robert W. Murray Lecture
Distinguished Alumni Lecture
-
Departmental News
News Archive
-
Facilities
High Field NMR Facility
MIST Lab
X-Ray Diffraction Facility
-
Alumni Interests
UMSL Chemists
Important Dates
Distinguished Alumni
Alumni Lecturers
-
Contact Information
|
|
Professor Dupureur received her B.S. degree from Southwest Missouri State University, and her Ph.D. from Ohio State University. She joined the UMSL Chemistry faculty in 2001. She held a faculty position at Texas A&M following a postdoctoral fellowship at the Caltech. She served as Department Chair Jan 2016 - July 2019. |
cdup@umsl.edu |
Research Interests
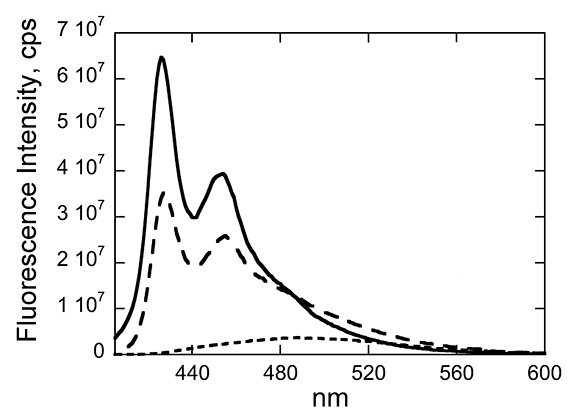
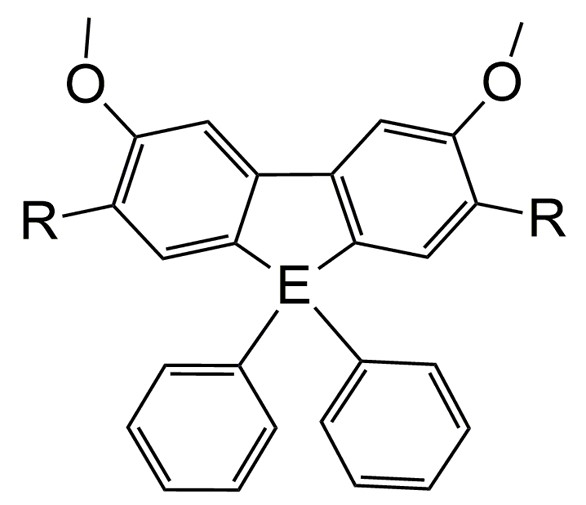
My group is interested in structure-function relationships, which is how the structure of a molecule dictates its biological activity. Our approach is biophysical, with an emphasis on spectroscopy. Over the years, we have worked with a number of other research groups on SAR (structure-activity relationships) in natural products and their analogs toward enzyme inhibition and drug-DNA interactions. Our most recent projects involve biological applications of metallafluorenes. These compounds have an affinity for surfactants and biological membranes, which suggest exciting applications that include fluorescence microscopy. Those working in the group master important instrumental and data analysis skills.

Recent Publications
″Tuning emission of luminescent 2-7 disubstituted sila-and germafluorenes with –(trifluoromethyl)phenyl, -(malononitrile)phenyl, and -nitrobenzene substituents,″ S. Jarret-Noland, T. Bandrowsky, J. Rajomoni, W. McConnell, J. Wilking and C. M. Dupureur, J. Organomet. Chem, 2024, 1005, 12297.
″Efficient production of fluorophore-labeled CC chemokines for biophysical studies using recombinant enterokinase and recombinant sortase,″ W. Guan, N. Zhang, A. Bains, M. Sadqi, C. M. Dupureur, P. J. LiWang, Biopolymers 2023, e23557. https://doi.org/10.1002/bip.23557
″Solvatochromic Behavior of 2,7-Disubstituted Sila-and Germafluorenes,″ S. J. Jarrett-Noland, W. McConnell, J. Braddock-Wilking and C. M. Dupureur, Chemosensors 2023, 11, 160.
"Size matters: DNA binding site kinetics as a function of polyamide size," J.Niederschulte, Y. Song, J. K. Bashkin and C. M. Dupureur, Biochimie, 2022, 199, 123.
″Group 14 Metallafluorenes as Sensitive Luminescent Probes of Surfactants in Aqueous Solution,″ H. J. Spikes, S. J. Jarrett-Noland, S. M. Germann, J. Braddock-Wilking and C. M. Dupureur, J. Fluoresc. 2021, 31, 916
″DNA binding site kinetics of a large antiviral polyamide,″ J. Niederschulte, Y. Song, A. H. Park, J. K. Bashkin and C. M. Dupureur, Biochemie, 2021, 185, 146.
″Synthesis of Luminescent 2-7 Disubstituted Silafluorenes with alkynyl-carbazole, -phenanthrene, and -benzaldehyde substituents,″ S. Germann, S. J. Jarrett, C. M. Dupureur, N. P. Rath, E. Gallaher and. Janet Braddock-Wilking, J. Organomet. Chem., 2020, 927, 121514.
″DNA binding thermodynamics and site stoichiometry as a function of polyamide size″, Y. Song, J. Niederschulte, K. N. Bales, A. H. Park, J. K. Bashkin and C. M. Dupureur, Biochimie 2019, 165, 170
″Thermodynamics and site stoichiometry of DNA binding by a large antiviral hairpin polyamide,″ Y. Song, J. Niederschulte, K. N. Bales, J. K. Bashkin and C. M. Dupureur, Biochimie, 2019, 157, 149.
″Biophysical and computational studies of the vCCI:vMIP-II complex,″ A. F. Nguyen, N.-W. Kuo, L. J. Showalter, R. Ramos, C. M.; Dupureur, M. E. Colvin and P. J. LiWang, Int. J. Mol. Sci. 2017,18, 1778.
″Stereospecific cholinesterase inhibition by O,S-diethylphenylphosphonothioate,″ A. Mohammed, J. Sneathen, S. G. Frojen, L. Kuo and C. M. Dupureur, Bioorg. & Med. Chem. 2017, 25, 3058. 8.
″Improved Antiviral Activity of a Polyamide Against High-Risk Human Papillomavirus Via N-Terminal Guanidinium Substitution,″ C. H. Castaneda, J. M. Scuderi, T. G. Edwards, G. D. Harris, C. M. Dupureur, K. J. Koeller, C. Fisher and J. K. Bashkin, MedChemComm. 2016, 7, 2076.
″Interactions of two large antiviral polyamides with the long control region of HPV16,″ E. Vasilieva, J. Niedershulte, Y. Song, J. K. Bashkin and C. M. Dupureur, Biochemie, 2016, 127, 103.
″Synthesis and comparison of the biological activity of monocyclic phosphonate, difluorophosphonate and phosphate analogs of the natural AChE inhibitor cyclophostin,″ B. P. Martin, E. Vasilieva, C. M. Dupureur and C. D. Spilling, Bioorg. & Med. Chem. 2015, 23, 7529.
“Rat hormone sensitive lipase inhibition by cyclopostins and their analogs”, E. Vasilieva, S. Dutta, R. K. Malla, B. P. Martin, C. D. Spilling and C. M. Dupureur, Bioorganic & Med. Chem. 2015, 23, 944.
