-
About Us
Keith J. Stine, Chair
Mission Statement
Degree Offerings
Department Photos
Outreach Activities
-
Undergraduate Studies
Undergraduate Degrees
Undergraduate Program Advising
Undergraduate Scholarships and Awards
Research for Undergraduates
Resources
-
Graduate Studies
Graduate Program Overview
Graduate Degrees
Application to Graduate Program
Graduate Awards
Graduate Program Contact Information
-
Faculty
Faculty Contact Information
Inorganic
Organic
Analytical/Physical
Biochemistry
Chemistry Faculty Awards
-
Staff
-
Seminar Programs
Archive
Graduate Student Seminars
Robert W. Murray Lecture
Distinguished Alumni Lecture
-
Departmental News
News Archive
-
Facilities
High Field NMR Facility
MIST Lab
X-Ray Diffraction Facility
-
Alumni Interests
UMSL Chemists
Important Dates
Distinguished Alumni
Alumni Lecturers
-
Contact Information
|
|
Professor Braddock-Wilking received her B.A. degree from the University of Missouri-St. Louis and her Ph.D. from Washington University. She joined the UMSL faculty in 1993 following postdoctoral fellowships at Harvard University and Mallinckrodt Medical, Inc. She retired in 2021 and assumed Emeritus status. |
wilkingj@umsl.edu |
Research Interests
Dr. Braddock-Wilking’s research focuses on the synthesis, characterization, and reactivity of compounds containing heavier Group 14 elements (E = Si, Ge). A major area of interest involves the chemistry of heterocyclopentadienes containing Group 14 elements, also known as metalloles (Figure 1), and related metallafluorenes and germafluoresceins (Figure 1). These heavy Group 14 molecules are known to exhibit unusual optoelectronic properties and high electron affinity and mobility and thus have potential application as components for electronic devices such as OLEDs and as chemical and biological sensors. Many of these molecules are weakly emissive in solution but show enhanced emission in the solid state due to the phenomenon of aggregation induced emission rendering them suitable for solid-state applications.
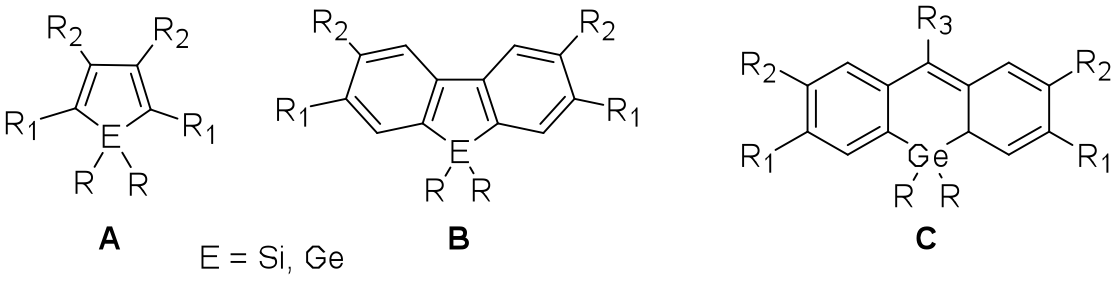 Fig. 1. General metallole and metallafluorene structures (M = Si, Ge)
Fig. 1. General metallole and metallafluorene structures (M = Si, Ge)
The research group is studying the structure-property relationship of these luminescent Group 14 molecules by exploring synthetic pathways that will allow for the incorporation of a variety of p-conjugated organic groups on the ring system enabling a range of emission colors (blue to red) and to promote aggregation-induced emission in the solid state. Figure 2 shows the molecular structure of an exceptionally stable germafluorene prepared in our laboratory that exhibits strong blue emission in solution and the solid state. The phenyl groups at the germanium center provide steric crowding that prevents close contact of adjacent molecules preventing aggregation-induced quenching which is commonly observed in the solid state with luminescent planar aromatic organic molecules.
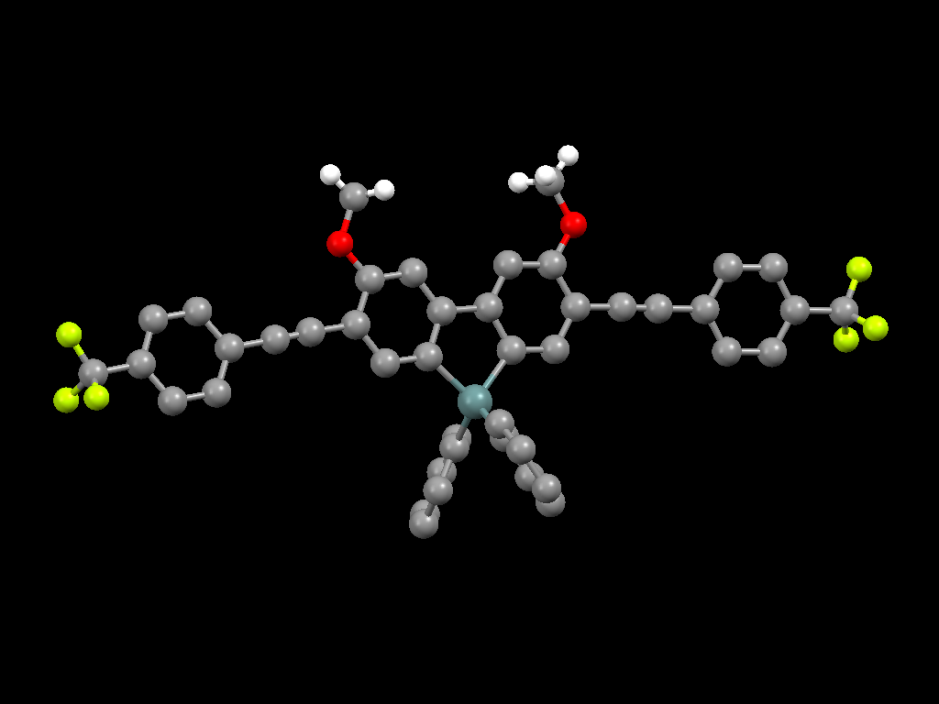 Fig. 2 Molecular structure of a germafluorene.
Fig. 2 Molecular structure of a germafluorene.
They are also investigating the synthesis of related systems that incorporate linker groups that can bind to transition-metal centers for the preparation of novel fluorescent metal containing complexes as well as for applications in chemical sensing. Figure 3 illustrates the fluorescence and Figure 4 shows related structures which are being examined with different chelating phosphines and nitrogen-based donors.

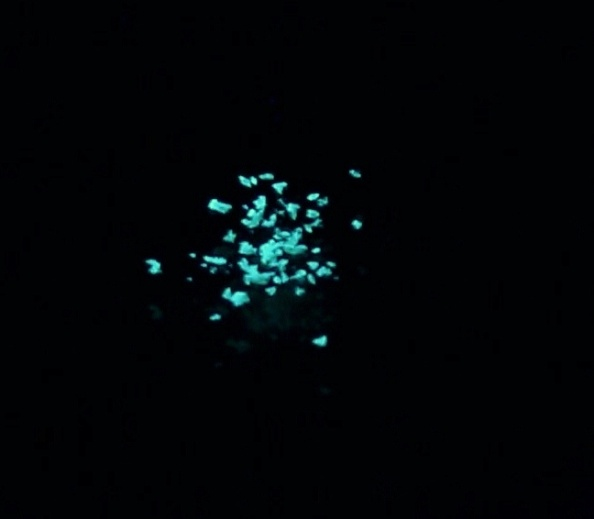 Fig. 3. Solid state fluorescence of a germafluorene on a tlc plate (top) and as crystals (bottom).
Fig. 3. Solid state fluorescence of a germafluorene on a tlc plate (top) and as crystals (bottom).
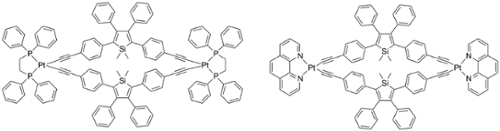 Fig. 4. Luminescent diplatinum macrocycles containing alkynyl-linked siloles.
Fig. 4. Luminescent diplatinum macrocycles containing alkynyl-linked siloles.
Selected Publications
″Tuning emission of luminescent 2-7 disubstituted sila-and germafluorenes with –(trifluoromethyl)phenyl, -(malononitrile)phenyl, and -nitrobenzene substituents,″ S. Jarret-Noland, T. Bandrowsky, J. Rajomoni, W. McConnell, J. Wilking and C. M. Dupureur, J. Organomet. Chem, 2024, 1005, 12297.
″Solvatochromic Behavior of 2,7-Disubstituted Sila-and Germafluorenes,″ S. J. Jarrett-Noland, W. McConnell, J. Braddock-Wilking and C. M. Dupureur, Chemosensors 2023, 11, 160.
″Group 14 Metallafluorenes as Sensitive Luminescent Probes of Surfactants in Aqueous Solution,″ H. J. Spikes, S. J. Jarrett-Noland, S. M. Germann, J. Braddock-Wilking and C. M. Dupureur, J. Fluoresc. 2021, 31, 916
″Synthesis of Luminescent 2-7 Disubstituted Silafluorenes with alkynyl-carbazole, -phenanthrene, and -benzaldehyde substituents,″ S. Germann, S. J. Jarrett, C. M. Dupureur, N. P. Rath, E. Gallaher and. Janet Braddock-Wilking, J. Organomet. Chem., 2020, 927, 121514.
″Luminescent 2,7-disubstituted germafluorenes", D. W. Hammerstroem, J. Braddock-Wilking and N. P. Rath, J. Organomet. Chem. 2017, 830, 196
"Synthesis and characterization of luminescent 2,7-disubstituted silafluorenes,″ D. W. Hammerstroem, J. Braddock-Wilking and N. P. Rath, J. Organomet. Chem. 2016, 813, 110
