-
About Us
Keith J. Stine, Chair
Mission Statement
Degree Offerings
Department Photos
Outreach Activities
-
Undergraduate Studies
Undergraduate Degrees
Undergraduate Program Advising
Undergraduate Scholarships and Awards
Research for Undergraduates
Resources
-
Graduate Studies
Graduate Program Overview
Graduate Degrees
Application to Graduate Program
Graduate Awards
Graduate Program Contact Information
-
Faculty
Faculty Contact Information
Inorganic
Organic
Analytical/Physical
Biochemistry
Chemistry Faculty Awards
-
Staff
-
Seminar Programs
Archive
Graduate Student Seminars
Robert W. Murray Lecture
Distinguished Alumni Lecture
-
Departmental News
News Archive
-
Facilities
High Field NMR Facility
MIST Lab
X-Ray Diffraction Facility
-
Alumni Interests
UMSL Chemists
Important Dates
Distinguished Alumni
Alumni Lecturers
-
Contact Information
 |
Dr. Hamper received his B.A. from Kalamazoo College and his Ph.D from the University of Illinois-Urbana. He began with Monsanto in St. Louis, and later joined G.D. Searle to pursue discovery medicinal chemistry. This led to research assignments in Pharmacia and Pfizer following a number of industry mergers/acquisitions. In 2010, he joined the faculty at UMSL and is now Associate Teaching Professor. |
hamperb@umsl.edu |
Research Interests
Our lab investigates continuous flow chemistry for the preparation of biologically relevant target molecules and improvement of reaction processes. Chemical reactions in continuous flow processes are inherently more efficient than batch processes, and can lead to advantages based on green chemistry principles. Monoliths and functionalized polymeric beads incorporating specifically designed organic molecules are prepared and evaluated for catalysis of reactions and selective absorption of solute molecules in flow devices. We employ a number of spectroscopic and chromatographic techniques to determine the nature of intermolecular interactions. This allows optimal selection of candidate molecules for incorporation into a larger molecular superstructure such as silica monoliths or polymeric materials.
Direct Uncatalyzed Amination of 2-Chloropyridine using a Flow Reactor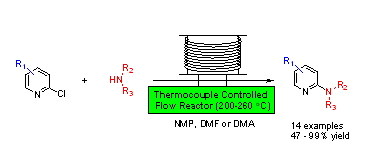 Chloropyridines are efficiently converted to 2-aminopyridines by uncatalyzed nucleophilic aromatic substitution (SNAr) in NMP, DMF or DMA using a continuous flow reactor. A variety of secondary amines undergo SNAr with both electron rich and electron deficient 2-chloropyridines to afford 2-aminopyridines in good to excellent yield. The flow reactor, which provides a short reaction time and high temperatures up to 300 oC, can overcome the activation barrier for reactions with unactivated substrates. Short reaction times result in fewer side products and can afford milligram to multi-gram quantities of product using continuous flow.
Chloropyridines are efficiently converted to 2-aminopyridines by uncatalyzed nucleophilic aromatic substitution (SNAr) in NMP, DMF or DMA using a continuous flow reactor. A variety of secondary amines undergo SNAr with both electron rich and electron deficient 2-chloropyridines to afford 2-aminopyridines in good to excellent yield. The flow reactor, which provides a short reaction time and high temperatures up to 300 oC, can overcome the activation barrier for reactions with unactivated substrates. Short reaction times result in fewer side products and can afford milligram to multi-gram quantities of product using continuous flow.
Beer Brewing – Chemical & Biochemical Principles
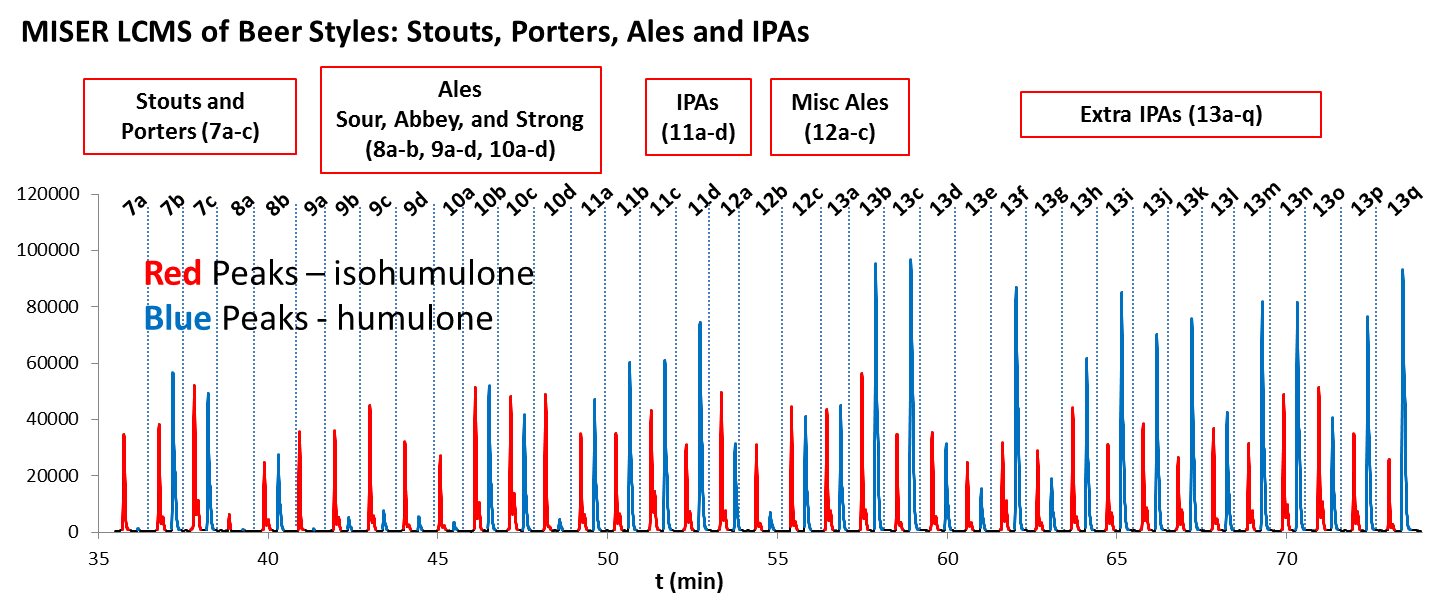
We developed an entirely new course offering for non-science majors, CHEM 1021: Beer Brewing – Chemical & Biochemical Principles. As an extension of CHEM 1021, we initiated a research project for analysis of hops derived, bittering agents in beer. Using MISER (multiple injections in a single experimental run) extracted ion LC-MS, we were able to obtain humulone-isohumulone profiles for large collections of beer samples. Rapid analysis of a group of 70 different beers in 74 minutes provided a comparative assessment of the amount of humulones (a-acids) and the isomerized isohumulones (iso-a-acids) present in an equivalent time of one minute per sample. The MISER LCMS approach allows for high-throughput analysis and a significant time savings, providing opportunities for increasing analysis sampling in production or a simple, rapid chemical analysis to accompany educational or classroom instruction.
Selected Recent Publications
″Liquid Chromatography–Mass Spectrometry (LC–MS) Analysis of Hop-Derived Humulone and Isohumulone Constituents in Beer: The Bitter Truth of Hops Utilization During Brewing″, B. C. Hamper, N. Viriyasiri, A. Boland, L. Espinosa, H. J. Campbell, K. Driesner and M. McKeever. LCGC North America, 2021, 39, 329
"Rapid LC-MS Analysis of Hop-Derived Humulone and Isohumulone Constituents in Beer", B. C. Hamper, N. Viriyasiri, A. Boland, L. Espinosa, H. Campbell and M. McKeever, J. CHEM. EDUC. 2020, 97, 1289.
″Introducing Nonscience Majors to Science Literacy via a Laboratory and Lecture Beer Brewing Course,″ B. C. Hamper and J. W. Meisel, J. CHEM. EDUC. 2020, 97, 1289.
″A Practical Gram-Scale Synthesis of Acrylohyd.roxamic Acid,″ B. C. Hamper, B. T. Sullivan, N. P. Rath and C. D. Spilling, Synthesis, 2017, 49, 355
″Rapid Determination of Humulones and Isohumulones in Beers Using MISER LC-MS Analysis,″ B. C. Hamper, K. Zawatzky, V. Zhang, and C. J. Welch, J. Am. Soc. Brew. Chem. 2017, 75, 333
″Enantioseparation of α-Hydroxyallylphosphonates and Phosphonoallylic Carbonate Derivatives on Chiral Stationary Phases Using Sequential UV, Polarimetric, and Refractive Index Detection,″ B. C. Hamper, M. P. Mannino, M. E. Mueller, L. T. Harrison and C. D. Spilling, Chirality, 2016, 28, 656.
″Discovery of N-(4-Fluoro-3-methoxy-benzyl)-6-(2-(((2S,5R)-5-(hydroxy-meth-yl)-1,4-dioxan-2-yl)methyl)-2H-tetrazol-5-yl)-2-methyl-pyrimidine-4-carbox-amide. A Highly Selective and Orally Bioavailable Matrix Metalloproteinase-13 Inhibitor for the Potential Treatment of Osteoarthritis,″ P. G. Ruminski, M. Massa, J, Strohbach, C. E. Hanau, M. Schmidt, J. A. Scholten, T. R. Fletcher, B. C. Hamper, Bruce C.; J. N. Carroll, H. S. Shieh, N. Caspers, B. Collins, M. Grapperhaus, K. E. Palmquist, J. Collins, J. E. Baldus, J. Hitchcock, H. P. Kleine et al. J. Med. Chem., 2016, 59, 313.
″Support of academic synthetic chemistry using separation technologies from the pharmaceutical industry,″ E. L. Regalado, M. C. Kozlowski, J. M. Curto, T. Ritter, M. G. Campbell, A. R. Mazzotti, B. C. Hamper, C. D. Spilling, M. P. Mannino, L. Wan, J-Q. Yu, J. Liu and C. J. Welch. Org. & Biomol Chem. 2014, 12, 2161
″A Practical and Scalable Synthesis of (S)- and (R)-1-(Dimethoxyphosphoryl)allyl Methyl Carbonates, ″ S. Roy, N. Sutivisedsak, B. C. Hamper, A. M. Lyss and C. D. Spilling, Synthesis 2014, 46, 3669
"Stucture-based parallel medincinal chemistry approach to improve metabolic stability of benzopyran COX-2 inhibitors," L. Xing, B. Hamper, T. R. Fletcher, J. M. Wendling, J. Carter, J. K. Gierse and S. Liao, Bioorg. Med. Chem. Lett. 2011, 21, 993
"The novel benzopyran class of selective cyclooxygenase-2 inhibitors. Part 3: The three microdose candidates," J. L. Wang, K. Aston, D. Lindburg, C. Ludwig, A. E. Hallinan, F. Koszyk, B. Hamper, D. Brown, M. Graneto, J. Talley, T. Maziasz, J. Masferrer and J. Carter, Bioorg. Med. Chem. Lett. 2010, 20, 7164-7168.
"The novel benzopyran class of selective cyclooxygenase-2 inhibitors. Part 2: The second clinical candidate having a shorter and favorable human half-life",J L. Wang, D. Lindburg, M. J. Graneto, J. Springer, J. Rogier, B. Hamper, S. Liao, J. L. Pawlitz, R. G. Kurumbail, T. Maziasz, J. J. Talley, J. R. Kiefer and J. Carter, Bioorg. Med. Chem. Lett. 2010, 20, 7159.
"Discovery of pyridine-4-yl -2H-tetrazole as a novel scaffold to identify highly selective matrix metalloproteinase-13 inhibitors for the treatment of osteoarthritis," M. E. Schnute, P. M. O’Brien, J. Nahra, M. Morris, W. H. Roark, C. E. Hanau, P. G. Ruminski, J. A. Scholten, T. R. Fletcher, B. C. Hamper, J. N. Carroll, W. C. Patt, H. S. Shieh, B. Collins, A. G. Pavlovsky, K. E. Palmquist, K. W. Aston, J. Hitchcock, M. D. Rogers, J. McDonald, A. R. Johnson, G. E. Munie, A. J. Wittwer, C.-F. Man, S. L. Settle, O. Nemirovskiy, L. E. Vickery, A. Agawal, R. D. Dyer and T. Sunyer, Bioorg. Med. Chem. Lett. 2010, 20, 576.
"Discovery of an Oral Potent Selective Inhibitor of Hematopoietic Prostaglandin D Synthase HPGDS , C. P. Carron, J. I. Trujillo, K. L. Olson, W. Huang, B. C. Hamper, T. Dice, B. E. Neal, M. J. Pelc, J. E. Day, D. C. Rohrer, J. R. Kiefer, J. B. Moon, B. A. Schweitzer, T. D. Blake, S. R. Turner, R. Woerndle, B. L. Case, C. P. Bono, V. M. Dilworth, C. L. Funckes-Shippy, B. L. Hood, G. M. Jerome, C. M. Kornmeier, M. R. Radabaugh, M. L. Williams, M. S. Davies, C. D. Wegner, D. J. Welsch, W. M. Abraham, C. J. Warren, M. E. Dowty, F. Hua, A. Zutshi, J. Z. Yang and A. Thorarensen," ACS Med. Chem. Lett. 2010, 1, 59.